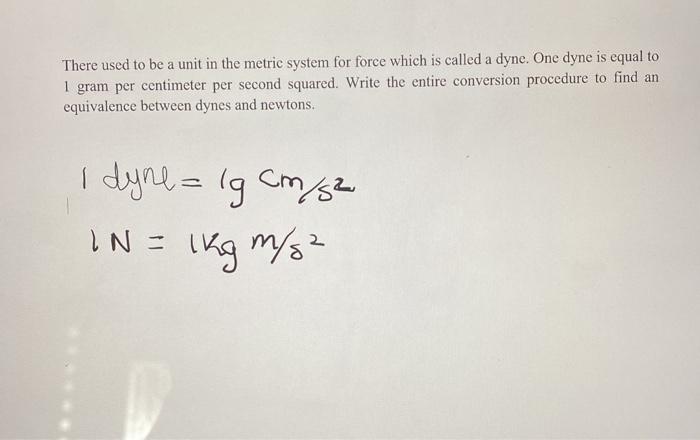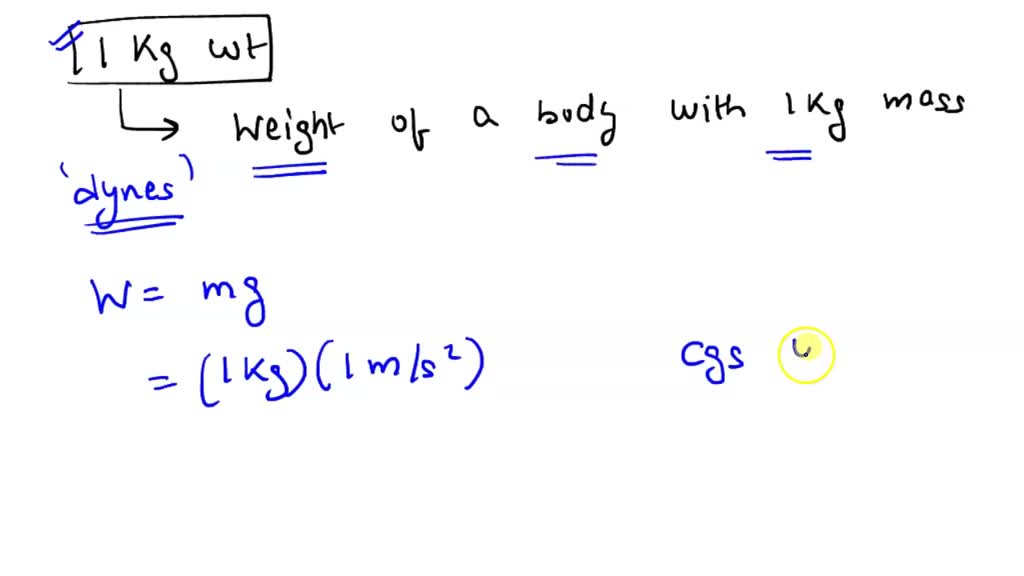If at a pressure of 10^6 dyne/cm^2 , one gram mole of nitrogen occupies 2 × 10^4 c.c. volume, then the average energy of nitrogen molecules in erg is :
ACCU DYNE TEST ™ Surface Tension Test Fluids formulated per ASTM Standard D2578 and ISO 8296 from 100% reagent grade materials

Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download

SOLVED:If at a pressure of 10^6 dyne / cm^2, one gram mole of nitrogen occupies 2 ×10^4 c.c. volume, then the average energy of a nitrogen molecules in erg is : (Given: